The New Product--Nitric Acid
The company's new products hit the shelves.
Nitric acid (English name: Nitric acid), is a strong oxidizing, corrosive monic inorganic acid, is one of the six inorganic strong acids, but also an important chemical raw materials, chemical formula for HNO3, molecular weight of 63.01, its aqueous solution commonly known as nitrite or ammonia water. In industry, it can be used to make chemical fertilizers, pesticides, explosives, dyes, etc. In organic chemistry, the mixture of concentrated nitric acid and concentrated sulfuric acid is an important nitrating reagent. The danger symbol is O (Oxidizing agent) and C (Corrosive corrosive agent). The anhydride of nitric acid is nitrous pentoxide (N2O5).
Pure nitric acid is a colorless transparent liquid, concentrated nitric acid is a light yellow liquid (dissolved with nitrogen dioxide), under normal circumstances, it is a colorless transparent liquid, with asphyxiating irritating odor. The nitric acid content in concentrated nitric acid is about 68%, which is volatile and produces white fog in the air (the same as concentrated hydrochloric acid), which is a small drop of nitric acid formed by the combination of nitric acid vapor (generally the nitrogen dioxide decomposed from concentrated nitric acid) and water vapor. It is miscible with water. It forms an azeotrope with water. For dilute nitric acid, it is generally believed that the boundary between dilute concentration is 6mol/L, the concentration of commercially available ordinary reagent grade nitric acid is about 68%, while the concentration of industrial grade concentrated nitric acid is 98%, and the concentration of usually fuming nitric acid is about 98%.
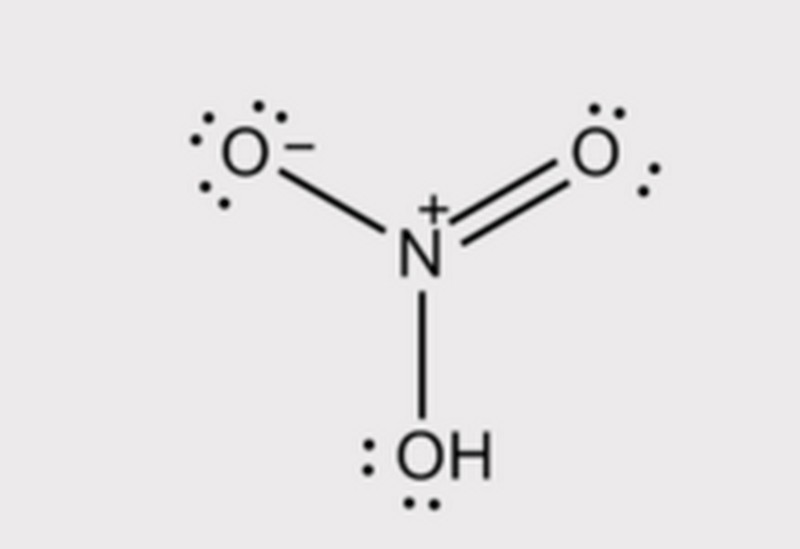
The nitric acid molecule is a planar covalent molecule, the central nitrogen atom is sp2 hybridized, and a p orbital not involved in hybridization forms a three-center four-electron bond with the two terminal oxygen [1]. The hydroxy hydrogen in nitric acid forms intramolecular hydrogen bond with the non-hydroxylated oxygen atom, which is the main reason why nitric acid is less acidic than sulfuric acid and hydrochloric acid, and the melting boiling point is lower than the first two.
Under normal circumstances, nitric acid can be completely ionized in an aqueous solution, producing a large number of hydrogen ions:
Nitric acid, as the highest (+5) hydrate of nitrogen, is very acidic.